The Indian pharmaceutical company Cipla has announced that they received regulatory approval by the drug controller general of India for launching the Favipiravir in the Country which will be sold under the brand name Ciplenza for restricted emergency use for the treatment of Corona Virus patients. ( Covid 19 patients )
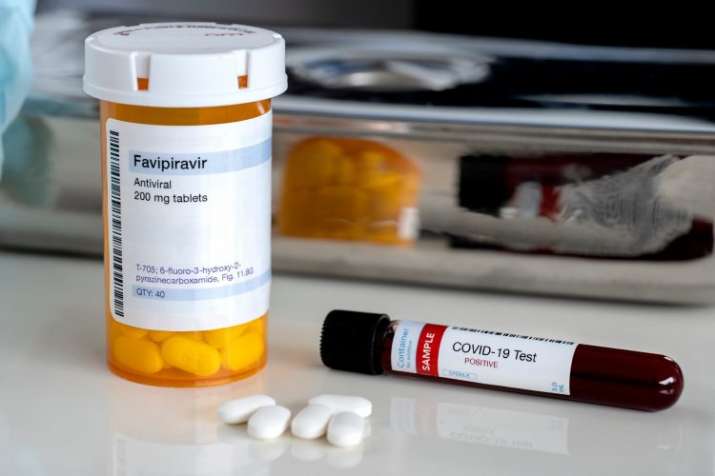 Cipla has announced that as a part of its efforts to meet the demand for the medicine, the commercial launch of the Ciplenza will take place in first week of August 2020 and they are charging Rs. 68 per tablet.
Cipla has announced that as a part of its efforts to meet the demand for the medicine, the commercial launch of the Ciplenza will take place in first week of August 2020 and they are charging Rs. 68 per tablet.
In order to ensure fair distribution of the drug, supplies will be undertaken through hospital channels and via open channels. Priority would be given for places of high burden of Covid-19 cases.
The drug has been jointly developed by Cipla and CSIR-Indian Institute of Chemical Technology. Favipiravir is an off patent, oral antiviral drug, originally discovered by Fuji Pharma in Japan and showed promising results in clinical trials for treatment of Covid patients especially in mild and moderate cases.
Director of CSIT-IICT Sri S,Chandrasekhar was quoted by news agency PTI as saying that the technology transferred to Cipla is very efficient and affordable allowing the company to make large quantities of the drug within a short span of time.
So it is a good news that the drug will be released in Indian market very soon.
 Cipla has announced that as a part of its efforts to meet the demand for the medicine, the commercial launch of the Ciplenza will take place in first week of August 2020 and they are charging Rs. 68 per tablet.
Cipla has announced that as a part of its efforts to meet the demand for the medicine, the commercial launch of the Ciplenza will take place in first week of August 2020 and they are charging Rs. 68 per tablet.In order to ensure fair distribution of the drug, supplies will be undertaken through hospital channels and via open channels. Priority would be given for places of high burden of Covid-19 cases.
The drug has been jointly developed by Cipla and CSIR-Indian Institute of Chemical Technology. Favipiravir is an off patent, oral antiviral drug, originally discovered by Fuji Pharma in Japan and showed promising results in clinical trials for treatment of Covid patients especially in mild and moderate cases.
Director of CSIT-IICT Sri S,Chandrasekhar was quoted by news agency PTI as saying that the technology transferred to Cipla is very efficient and affordable allowing the company to make large quantities of the drug within a short span of time.
So it is a good news that the drug will be released in Indian market very soon.
No comments:
Post a Comment